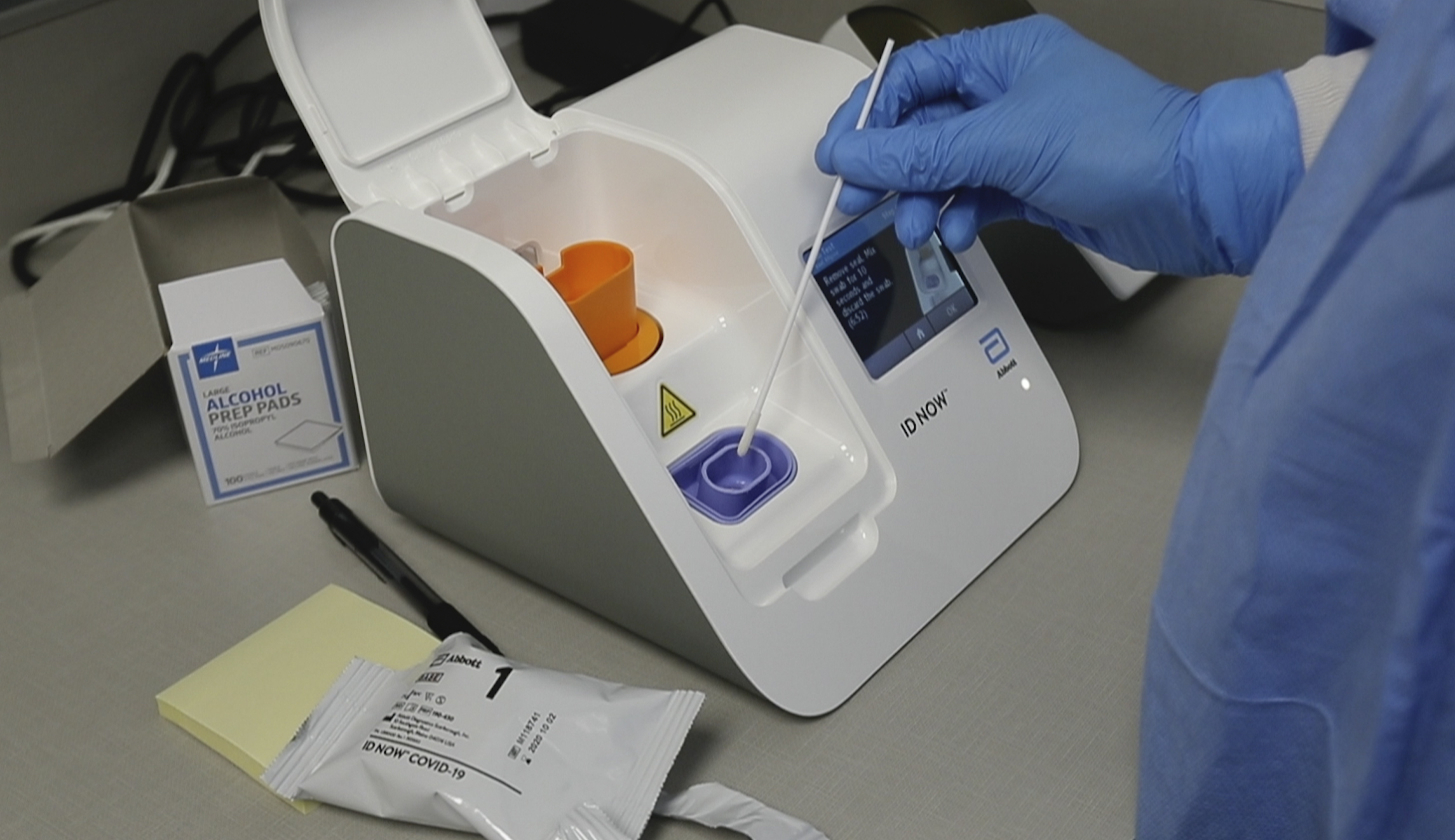
The Food and Drug Administration issued an alert about “possible accuracy problems” with the rapid coronavirus test used by the White House after studies questioned its accuracy.
A recent New York University study, which has not yet been peer-reviewed, found that Abbott’s ID NOW missed at least one-third of positive cases detected by a different test. The number of false negatives increased when using the currently recommended dry nasal swabs.
Recommended Stories
“This test can still be used and can correctly identify many positive cases in minutes. Negative results may need to be confirmed with a high-sensitivity authorized molecular test,” FDA official Tim Stenzel said Thursday.
However, Abbott disputed the latest findings, saying 1.8 million tests had been distributed with a reported rate of 0.02% false negatives.
“Once again, a study has been conducted using ID NOW in a manner that it’s not intended to be used,” said Scott Stoffel, divisional vice president for external communications. “It’s unclear if the samples were tested correctly, and we’re further evaluating these results.”
The White House is using the rapid test to test staff, reporters, and others who frequently come in contact with the president and vice president.
The FDA said it was working with Abbott to inform users that they may need another test if they were given negative results but had symptoms of the coronavirus.